Standard molar entropy table4/8/2024 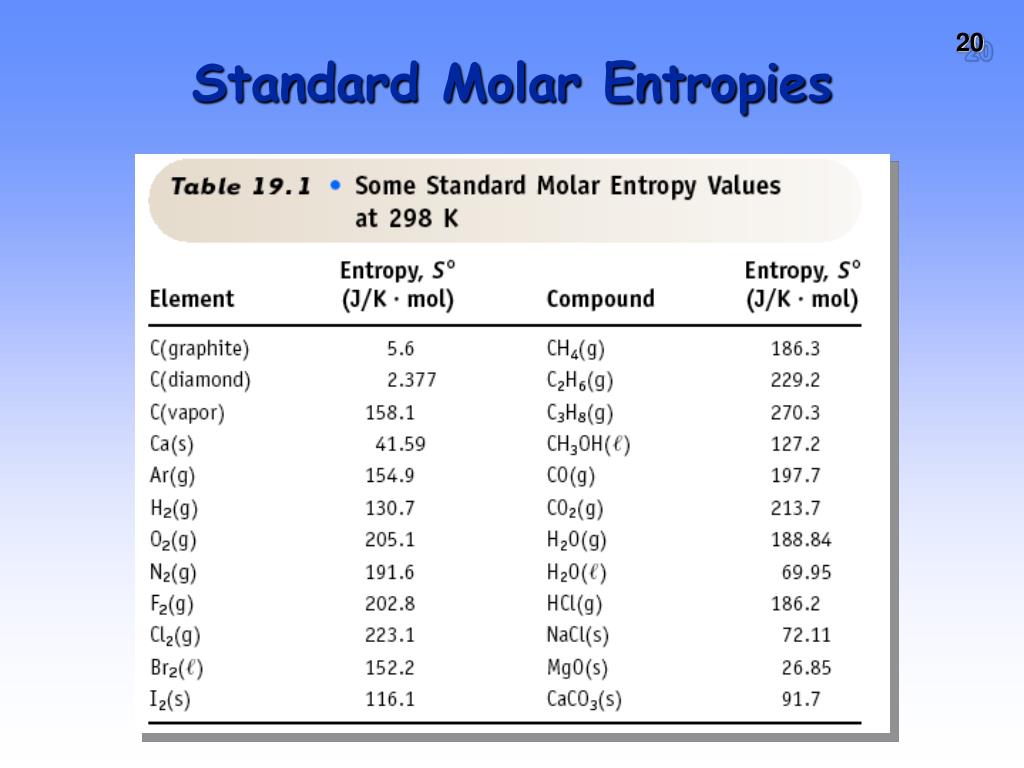 
15 K, I = 0 ) of species are of more interest because it may be possible to interpret their values and use them to estimate S m ∘ values for species for which Δ f H j ∘ are unknown. However, standard molar entropies S m ∘ ( 298. With the database BasicBiochemData2 and recent extensions, this can be done for about fifty reactants. 15 K, I = 0 ) of the species of a reactant are known, the standard entropies of formation Δ f S j ∘ ( 298. 15 K, I = 0 ) and standard enthalpies of formation Δ f H j ∘ ( 298. When the standard Gibbs energies of formation Δ f G j ∘ ( 298. The standard transformed entropy of reaction has received less attention, but it provides almost all of the pH dependence of the apparent equilibrium constant. The standard transformed enthalpy of reaction yields the heat of reaction. The standard transformed Gibbs energy of reaction provides the criterion for spontaneous change and equilibrium for an enzyme-catalyzed reaction at specified temperature, pressure, and pH.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
